Abstract
Purpose
To compare docetaxel-based doublet with single-agent docetaxel as second-line treatment in non-small-cell lung cancer (NSCLC).
Methods
We systematically searched for randomized clinical trials that compared docetaxel-based doublet with single-agent docetaxel in patients with histologically proven non-small-cell lung cancer. The primary end point was overall survival (OS). Secondary end points were progression-free survival, overall response rate, 1-year survival rate, and grade 3 or 4 toxicity. Data were extracted from the studies by two independent reviewers. The meta-analysis was performed by Stata version 10.0 software (Stata Corporation, College Station, TX, USA).
Results
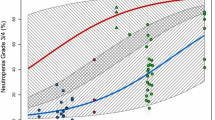
Eight randomized clinical trials (totally 2,126 patients) were eligible. Meta-analysis showed that there was significant improvement in PFS (HR 0.81, 95% CI 0.69–0.96, P = 0.013) and overall response rate (OR 1.42, 95% CI 1.13–1.80, P = 0.03) in docetaxel-based doublet group, compared with docetaxel alone, though the pooled HR for overall survival (HR 0.93, 95% CI 0.80–1.07, P = 0.308) showed no significant difference between the two groups. However, there were more incidences of grade 3 or 4 neutropenia (OR 1.2, 95% CI 1.00–1.45, P = 0.05), thrombocytopenia (OR 4.53, 95% CI 1.75–11.75, P = 0.002), and diarrhea (OR 1.78, 95% CI 1.16–2.74, P = 0.008) in docetaxel-based doublet group. With regard to the risk of grade 3 or 4 anemia (OR 1.95, 95% CI 0.62–6.17, P = 0.25), fatigue (OR 1.09, 95% CI 0.75–1.59, P = 0.66), and nausea and vomiting (OR 1.75, 95% CI 0.78–3.91, P = 0.17), there was no significant difference between the two groups.
Conclusions
This was the first meta-analysis of docetaxel-based doublet versus single-agent docetaxel as second-line therapy in the treatment of non-small-cell lung cancer. The results indicated that docetaxel-based doublet therapy did not gain any benefit in survival but significantly improved PFS and better ORR versus single-agent docetaxel. However, more incidences of grade 3 or 4 neutropenia, thrombocytopenia, and diarrhea were observed in docetaxel-based doublet group.






Similar content being viewed by others
References
Peto R, Chen ZM, Boreham J (1999) Tobacco—the growing epidemic. Nat Med 5:15–17
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ (2009) Cancer statisktics, 2009. CA Cancer J Clin 59:225–249
Shepherd FA (1993) Screening, diagnosis and staging of lung cancer. Curr Opin Oncol 5:310–322
Shepherd FA, Dancey J, Ramlau R, Mattson K, Gralla R, O’Rourke M, Levitan N, Gressot L, Vincent M, Burkes R, Coughlin S, Kim Y, Berille J (2000) Prospective randomized trial of docetaxel versus best supportive care in patients with non-small-cell lung cancer previously treated with platinum-based chemotherapy. J Clin Oncol 18:2095–2103
Fossella FV, DeVore R, Kerr RN, Mattson K, Gralla R, O’Rourke M, Levitan N, Gressot L, Vincent M, Burkes R, Coughlin S, Kim Y, Berille J (2000) Randomized phase III trial of docetaxel versus vinorelbine or ifosfamide in patients with advanced non-small-cell lung cancer previously treated with platinum-containing chemotherapy regimens: the TAX 320 Non-Small Cell Lung Cancer Study Group. J Clin Oncol 18:2354–2362
Dancey J, Shepherd FA, Gralla RJ, Kim YS (2004) Quality of life assessment of second-line docetaxel versus best supportive care in patients with non-small-cell lung cancer previously treated with platinum-based chemotherapy: results of a prospective randomized phase III trial. Lung Cancer 43:183–194
Delbaldo C, Michiels S, Syz N, Soria JC, Le Chevalier T, Pignon JP (2004) Benefits of adding a drug to a single-agent or a 2-agent chemotherapy regimen in advanced non-small-cell lung cancer: a meta-analysis. JAMA 292:470–484
Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L, Verweij J, Van Glabbeke M, van Oosterom AT, Christian MC, Gwyther SG (2000) New guidelines to evaluate the response to treatment in solid tumors: European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst 92:205–216
Parmar MK, Torri V, Stewart L (1998) Extracting summary statistics to perform meta-analyses of the published literature for survival endpoints. Stat Med 17:2815–2834
Zintzaras E, Ioannidis JP (2005) Heterogeneity testing in meta-analysis of genome searches. Genet Epidemiol 28:123–137
Yusuf S, Peto R, Jones DR, Collins R, Sleight P (1985) Beta blockade during and after myocardial infarction: an overview of the randomized trials. Prog Cardiovasc Dis 27:335–371
Begg CB, Mazumdar M (1994) Operating characteristics of a rank correlation test for publication bias. Biometrics 50:1088–1101
Moher D, Pham B, Jones A, Cook DJ, Jadad AR, Moher M, Tugwell P, Klassen TP (1998) Does quality of reports of randomized trials affect estimates of intervention efficacy reported in meta-analyses? Lancet 352:609–613
Herbst RS, Sun Y, Eberhardt WE, Germonpré P, Saijo N, Zhou C, Wang J, Li L, Kabbinavar F, Ichinose Y, Qin S, Zhang L, Biesma B, Heymach JV, Langmuir P, Kennedy SJ, Tada H, Johnson BE (2010) Vandetanib plus docetaxel versus docetaxel as second-line treatment for patients with advanced non-small-cell lung cancer (ZODIAC): a double-blind, randomised, phase 3 trial. Lancet Oncol 11:619–626
Gebbia V, Gridelli C, Verusio C, Frontini L, Aitini E, Daniele B, Gamucci T, Mancuso G, Di Maio M, Gallo C, Perrone F, Morabito A (2009) Weekly docetaxel vs. docetaxel-based combination chemotherapy as second-line treatment of advanced non-small-cell lung cancer patients. The DISTAL-2 randomized trial. Lung Cancer 63:251–258
Pectasides D, Pectasides M, Farmakis D (2005) Comparison of docetaxel and docetaxel–irinotecan combination as second-line chemotherapy in advanced non-small-cell lung cancer: a randomized phase II trial. Ann Oncol 16:294–299
Takeda K, Negoro S, Tamura T, Nishiwaki Y, Kudoh S, Yokota S, Matsui K, Semba H, Nakagawa K, Takada Y, Ando M, Shibata T, Saijo N (2009) Phase III trial of docetaxel plus gemcitabine versus docetaxel in second-line treatment for non-small-cell lung cancer: results of a Japan Clinical Oncology Group trial (JCOG0104). Ann Oncol 20:835–841
Pallis AG, Agelaki S, Agelidou A, Varthalitis I, Syrigos K, Kentepozidis N, Pavlakou G, Kotsakis A, Kontopodis E, Georgoulias V (2010) A randomized phase III study of the docetaxel/carboplatin combination versus docetaxel single-agent as second line treatment for patients with advanced/metastatic non-small cell lung cancer. BMC Cancer 10:633
Heymach JV, Johnson BE, Prager D, Csada E, Roubec J, Pesek M, Spásová I, Belani CP, Bodrogi I, Gadgeel S, Kennedy SJ, Hou J, Herbst RS (2007) Randomized, placebo-controlled phase II study of vandetanib plus docetaxel in previously treated non small-cell lung cancer. J Clin Oncol 25:4270–4277
Wachters FM, Groen HJ, Biesma B, Schramel FM, Postmus PE, Stigt JA, Smit EF (2005) A randomized phase II trial of docetaxel vs. docetaxel and irinotecan in patients with stage IIIb–IV non-small-cell lung cancer who failed first-line treatment. Br J Cancer 92:15–20
Segawa Y, Kiura K, Hotta K, Takigawa N, Tabata M, Matsuo K, Yoshioka H, Hayashi H, Kawai H, Aoe K, Maeda T, Ueoka H, Tanimoto M (2010) A randomized phase II study of a combination of docetaxel and S-1 versus docetaxel monotherapy in patients with non-small cell lung cancer previously treated with platinum-based chemotherapy: results of Okayama Lung Cancer Study Group (OLCSG) Trial 0503. J Thorac Oncol 5:1430–1434
Di Maio M, Chiodini P, Georgoulias V, Hatzidaki D, Takeda K, Wachters FM, Gebbia V, Smit EF, Morabito A, Gallo C, Perrone F, Gridelli C (2009) Meta-analysis of single-agent chemotherapy compared with combination chemotherapy as second-line treatment of advanced non-small-cell lung cancer. J Clin Oncol 27:1836–1843
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Qi, WX., Shen, Z. & Yao, Y. Meta-analysis of docetaxel-based doublet versus docetaxel alone as second-line treatment for advanced non-small-cell lung cancer. Cancer Chemother Pharmacol 69, 99–106 (2012). https://doi.org/10.1007/s00280-011-1678-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-011-1678-9